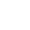Otu Cyano nwere ike ibuli elu na nnabata elektrọn siri ike, ya mere ọ nwere ike ịbanye n'ime protein ebumnuche iji mepụta njikọ hydrogen na ihe fọdụrụ amino acid dị mkpa na ebe ọrụ. N'otu oge ahụ, otu cyano bụ ahụ bioelectronic isosteric nke carbonyl, halogen na otu ọrụ ndị ọzọ, nke nwere ike ime ka mmekọrịta dị n'etiti obere molekul ọgwụ na protein ebumnuche dịkwuo mma, yabụ a na-eji ya nke ọma na mgbanwe nhazi nke ọgwụ na ọgwụ ahụhụ [1]. Ọgwụ ọgwụ cyano nke nwere ọgwụ gụnyere saxagliptin (Foto 1), verapamil, febuxostat, wdg; Ọgwụ ugbo gụnyere bromofenitrile, fipronil, fipronil na ndị ọzọ. Na mgbakwunye, ngwakọta cyano nwekwara uru dị mkpa n'ọhịa nke isi ísì, ihe ọrụ na ndị ọzọ. Dịka ọmụmaatụ, Citronitrile bụ isi nitrile ọhụrụ mba ụwa, 4-bromo-2,6-difluorobenzonitrile bụkwa ihe dị mkpa maka ịkwadebe ihe kristal mmiri mmiri. A pụrụ ịhụ na a na-eji ngwakọta cyano eme ihe n'ọtụtụ ebe n'ihi ihe pụrụ iche ha [2].
Otu Cyano nwere ike ibuli elu na nnabata elektrọn siri ike, ya mere ọ nwere ike ịbanye n'ime protein ebumnuche iji mepụta njikọ hydrogen na ihe fọdụrụ amino acid dị mkpa na ebe ọrụ. N'otu oge ahụ, otu cyano bụ ahụ bioelectronic isosteric nke carbonyl, halogen na otu ọrụ ndị ọzọ, nke nwere ike ime ka mmekọrịta dị n'etiti obere molekul ọgwụ na protein ebumnuche dịkwuo mma, yabụ a na-eji ya nke ọma na mgbanwe nhazi nke ọgwụ na ọgwụ ahụhụ [1]. Ọgwụ ọgwụ cyano nke nwere ọgwụ gụnyere saxagliptin (Foto 1), verapamil, febuxostat, wdg; Ọgwụ ugbo gụnyere bromofenitrile, fipronil, fipronil na ndị ọzọ. Na mgbakwunye, ngwakọta cyano nwekwara uru dị mkpa n'ọhịa nke isi ísì, ihe ọrụ na ndị ọzọ. Dịka ọmụmaatụ, Citronitrile bụ isi nitrile ọhụrụ mba ụwa, 4-bromo-2,6-difluorobenzonitrile bụkwa ihe dị mkpa maka ịkwadebe ihe kristal mmiri mmiri. A pụrụ ịhụ na a na-eji ngwakọta cyano eme ihe n'ọtụtụ ebe n'ihi ihe pụrụ iche ha [2].
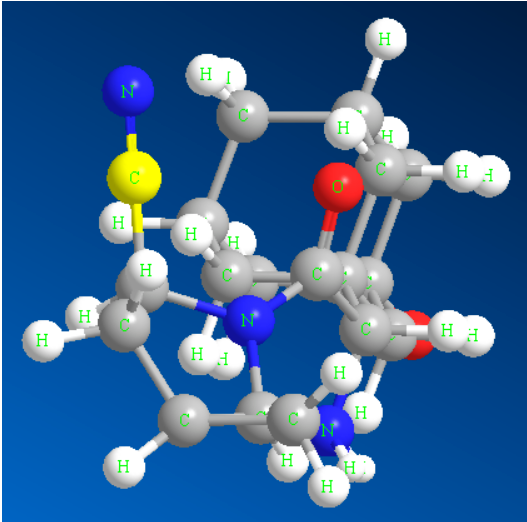
2.2 mmeghachi omume cyanidation electrophilic nke enol boride
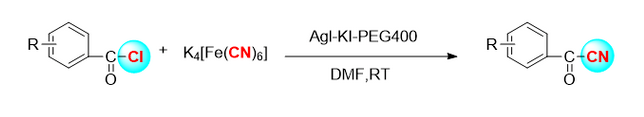
Ndị otu Kensuke Kiyokawa [4] jiri ihe ndị na-eme cyanide n-cyano-n-phenyl-p-toluenesulfonamide (NCTS) na p-toluenesulfonyl cyanide (tscn) mee ihe iji nweta cyanidation electrophilic dị elu nke ogige enol boron (Foto 3). Site na atụmatụ ọhụrụ a, ọtụtụ β-Acetonitrile dị iche iche, ma nwee ọtụtụ ihe ndị na-emepụta ihe.
2.3 mmeghachi omume nke ketones nke silico cyanide nke organic catalytic
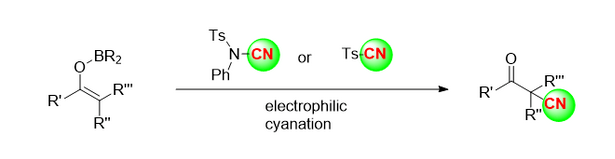
N'oge na-adịbeghị anya, ndị otu ndepụta Benjamin [5] kọrọ na akwụkwọ akụkọ Nature nke ọdịiche enantiomeric nke 2-butanone (Foto 4a) na mmeghachi omume cyanide asymmetric nke 2-butanone nwere enzymes, ihe na-akpali akpali organic na ihe na-akpali akpali metal transition, site na iji HCN ma ọ bụ tmscn dị ka ihe na-akpali akpali cyanide (Foto 4b). Site na tmscn dị ka ihe na-akpali akpali cyanide, 2-butanone na ọtụtụ ketones ndị ọzọ nwere mmeghachi omume silyl cyanide dị oke enantioselective n'okpuru ọnọdụ ihe na-akpali akpali nke idpi (Foto 4C).
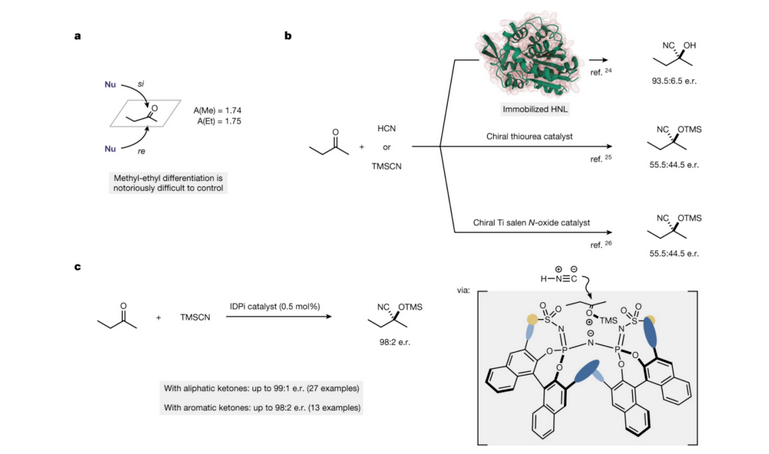
Foto nke 4 A, ọdịiche enantiomeric nke 2-butanone. b. Cyanidation asymmetric nke 2-butanone na enzymes, ihe ndị na-akpali akpali organic na ihe ndị na-akpali mgbanwe metal.
c. Idpi na-akpali mmeghachi omume silyl cyanide nke enantioselective nke 2-butanone na ọtụtụ ketones ndị ọzọ.
2.4 mbelata cyanidation nke aldehydes
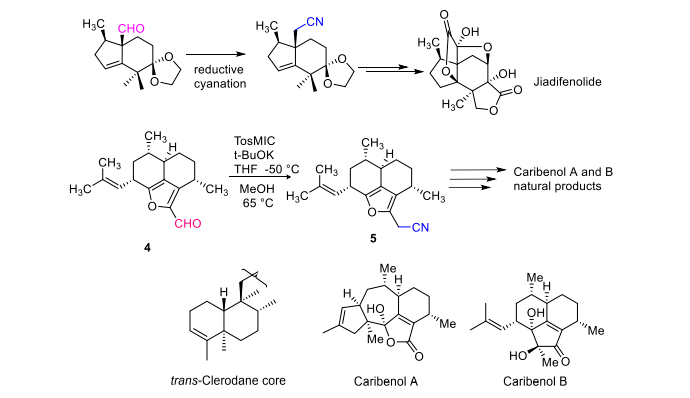
N'ime mmepụta nke ngwaahịa eke, a na-eji tosmic akwụkwọ ndụ akwụkwọ ndụ dị ka ihe na-eme ka ihe na-eme ka aldehydes ndị a na-egbochi sterically ka ọ bụrụ nitriles ngwa ngwa. A na-ejikwa usoro a ewebata atọm carbon ọzọ n'ime aldehydes na ketones. Usoro a nwere ihe bara uru na njikọ zuru oke nke jiadifenolide nke Enantiospecific ma bụrụ nzọụkwụ dị mkpa na njikọta nke ngwaahịa eke, dị ka njikọta nke ngwaahịa eke dịka clerodane, caribenol A na caribenol B [6] (Foto 5).
Mmeghachi omume cyanide electrochemical 2.5 nke amine organic
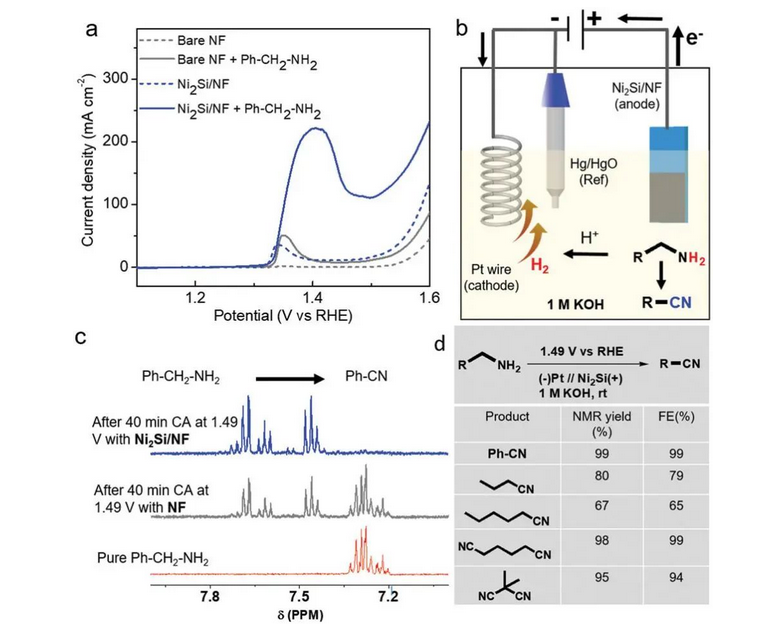
Dịka teknụzụ mmepụta ihe ndụ ndụ, a na-eji usoro mmepụta ihe electrochemical organic eme ihe n'ọtụtụ ebe n'ọtụtụ ebe mmepụta ihe organic. N'afọ ndị na-adịbeghị anya, ọtụtụ ndị nchọpụta elebala anya na ya. Ndị otu PrashanthW. Menezes [7] kọrọ n'oge na-adịbeghị anya na enwere ike itinye amine aromatic ma ọ bụ aliphatic amine ozugbo na ngwakọta cyano kwekọrọ na ngwọta KOH 1m (na-etinyeghị cyanide reagent) yana ikike mgbe niile nke 1.49vrhe site na iji ihe na-eme ka Ni2Si dị ọnụ ala, yana nnukwu mkpụrụ (Foto 6).
Nchịkọta 03
Cyanidation bụ mmeghachi omume dị oke mkpa nke ihe ndị sitere n'okike. Site n'echiche nke kemistri akwụkwọ ndụ akwụkwọ ndụ, a na-eji ihe ndị na-eme cyanide na-adịghị mma na gburugburu ebe obibi dochie ihe ndị na-eme cyanide ọdịnala na-egbu egbu na ndị na-emerụ ahụ, a na-ejikwa ụzọ ọhụrụ dị ka ihe na-enweghị ihe mgbaze, ihe na-abụghị catalytic na radieshon microwave iji gbasaa oke na omimi nke nyocha, iji mepụta nnukwu uru akụ na ụba, mmekọrịta mmadụ na ibe ya na gburugburu ebe obibi na mmepụta ụlọ ọrụ [8]. Site na ọganihu na-aga n'ihu nke nyocha sayensị, mmeghachi omume cyanide ga-etolite na mmepụta dị elu, akụ na ụba na kemịkalụ akwụkwọ ndụ akwụkwọ ndụ.
Oge ozi: Sep-07-2022